Acid stratification is a common cause of battery failure. In a stratified battery, the electrolyte is concentrated at the bottom and the top half of the cell has very little acid. Stratification takes place when the battery is kept with low charge (below 80%) and is never fully recharged. Short journeys that include the use of the windscreen wipers and electric heaters contribute to this phenomenon. Acid stratification reduces the general performance of the battery.
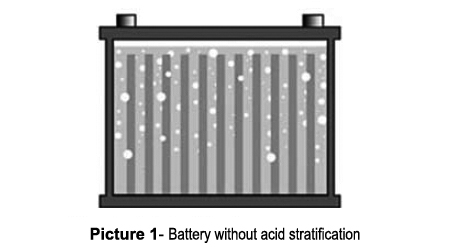
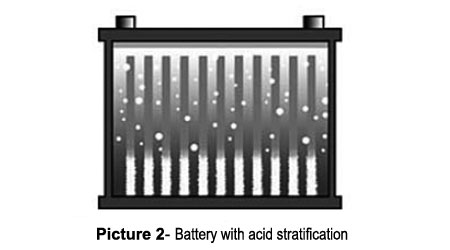
Picture 1 shows a normal battery in which the acid is distributed evenly from top to bottom. This battery has good performance because the correct concentration of acid is distributed evenly over the plates. Picture 2 shows a stratified battery in which the acid concentration is light at the top and heavy at the bottom. A light acid limits the plate activation, speeds up corrosion and reduces the performance. On the other hand, high acid concentration at the bottom artificially increases the open circuit voltage. The battery seems to be fully charged, but it delivers low starting power. High acid concentration also results in sulphation and further reduces the already low conductivity. If this condition goes undetected, it will ultimately result in battery failure.
Fully recharging or shaking the battery tends to correct the problem.